| International Journal of Clinical Pediatrics, ISSN 1927-1255 print, 1927-1263 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Int J Clin Pediatr and Elmer Press Inc |
| Journal website https://www.theijcp.org |
Case Report
Volume 11, Number 1, March 2022, pages 20-26
The Management of Influenza Virus-Induced Plastic Bronchitis in Pediatric Patients: A Case Report and Literature Review
Makoto Miyamotoa, c, Yasuhiro Yanagishitab, Asami Kureb, Satoru Hashimotob, Toshihiko Takiurab, Akira Hosakib, Kunimasa Yanb, Koichiro Saitoa
aDepartment of Otolaryngology-Head and Neck Surgery, Kyorin University School of Medicine, Mitaka, Tokyo, Japan
bDepartment of Pediatrics, Kyorin University School of Medicine, Mitaka, Tokyo, Japan
cCorresponding Author: Makoto Miyamoto, Department of Otolaryngology-Head and Neck Surgery, Kyorin University School of Medicine, Tokyo, Japan
Manuscript submitted January 22, 2022, accepted February 7, 2022, published online March 12, 2022
Short title: Influenza Virus-Induced Plastic Bronchitis
doi: https://doi.org/10.14740/ijcp476
| Abstract | ▴Top |
Although the incidence is very rare, plastic bronchitis (PB) could be a serious comorbid disease of virus infection in children, especially in those who have a history of bronchial asthma. We have recently experienced an influenza virus-induced PB who was successfully treated by endoscopic intervention under multidisciplinary approach in our institution. The purpose of this report is to confirm the role of otolaryngologists in the management of potentially lethal pediatric PB. A 7-year-old girl visited our institution (Kyorin University Hospital) and was hospitalized to the Pediatrics Department with worsening cough and chest pain. Chest radiography and computed tomography imaging showed a complete atelectasis of the left lung. Influenza virus testing proved the infection of influenza A in this patient, and she was treated by antiviral medication (peramivir hydrate), systemic steroid therapy and respiratory rehabilitation. However, her symptoms were worsening, and she was consulted to our department. Multiple clinical information suggested PB, and bronchoscopic evaluation/treatment was performed under general anesthesia using laryngeal mask 4 days after her hospitalization. Endoscopic intervention visualized the blockage of left main bronchus with highly viscous mucous, and her symptoms were improved by endoscopic suctioning of the mucus; and it was found that the inflammatory cells were eosinophil. Her pulmonary condition got better immediately after suctioning, and she was discharged 10 days after the intervention. Our report suggested the indispensable role of otolaryngologist for successful diagnosis and treatment of the influenza virus-induced PB in pediatric patients.
Keywords: Plastic bronchitis; Influenza virus-induced PB; Atelectasis; Endoscopic suctioning; Eosinophil
| Introduction | ▴Top |
Seasonal influenza is an acute respiratory infection caused by the influenza virus and is self-limited, and patients usually recover without complications. In Japan, an epidemic is observed every winter season, and in a typical case, symptoms suddenly develop with the upper respiratory tract, such as fever greater than 38 °C, rhinorrhea, and cough. In addition, symptoms are accompanied by muscle pain, vomiting, and diarrhea and then reach the lower respiratory tract, causing moist cough and gradual recovery. If dyspnea occurs while experiencing influenza and atelectasis is observed on chest radiography, plastic bronchitis (PB) is considered. PB is often reported during influenza pandemic [1].
In this report, a 7-year-old girl presented with respiratory distress after experiencing seasonal influenza A, and atelectasis of the left lung was observed on chest radiography and computed tomography (CT). She did not recover with conservative treatment, and we performed the suctioning of the mucous plug using flexible bronchoscopy. We described the clinical features, chest radiographic results, treatment, and role of bronchoscopy.
| Case Report | ▴Top |
A previous healthy 7-year-old girl had a severe cough 3 days previously and was provided a common cold medicine as the diagnosis for streptococcal infection in a pediatric clinic (clarithromysin 200 mg/day, L-carbocistein 750 mg/day, and ambroxol hydrochloride 45 mg/day). However, her condition did not improve, and her cough was worsened. Moreover, she developed left upper body pain, and was admitted to the Pediatric Department of our university hospital on Sunday. She lives with her parents and a younger brother, and there is no pet at home. She had no remarkable medical, family, psychosocial, birth, and developmental histories. She had no drug allergy, and no food allergy, but she had allergic rhinitis. She had received all vaccinations as scheduled in Japan Pediatric Society; however, she did not receive flu vaccination this winter.
Physical examination at hospital admission revealed respiratory distress, as indicated by tachypnea (respiratory rate, 28 breaths/min; peripheral oxygen saturation, 92% in room air). The patient’s breath sounds were poor in the left lung, and wheezing was heard in the right lung. Moreover, she had shoulder breathing and mild retractive breathing. Her temperature was 37.1 °C, heart rate was 116/min, and her blood pressure was 114/72 mm Hg. Upon cardiac auscultation, the rhythm was rapid but regular, no cardiac murmur was detected. There were no other findings on physical and neurological examination. The patient’s laboratory examinations were as follows: white blood cell count, 8,200/µL (neutrophils, 79.9%; eosinophils, 1.5%); C-reactive protein level, 0.10 mg/dL; and total immunoglobulin E, 282 IU/mL. The arterial blood gas analysis was as follows: pH, 7.399; pCO2, 37.2 mm Hg; pO2, 50.5 mm Hg; HCO3, 22.5 mm Hg; base excess, -1.5; and oxygen saturation, 85.2%. The rapid nasal swab influenza antigen test was negative for influenza A and B, and the anti-mycoplasma antibody test was negative (Table 1). Chest radiography revealed low permeability in the left lung field. Chest CT revealed decreased permeability in the left lung field (Fig. 1a-c). The patient was diagnosed with atelectasis of the left lobe and was admitted to the Pediatric Department.
 Click to view | Table 1. Laboratory Data on Admission |
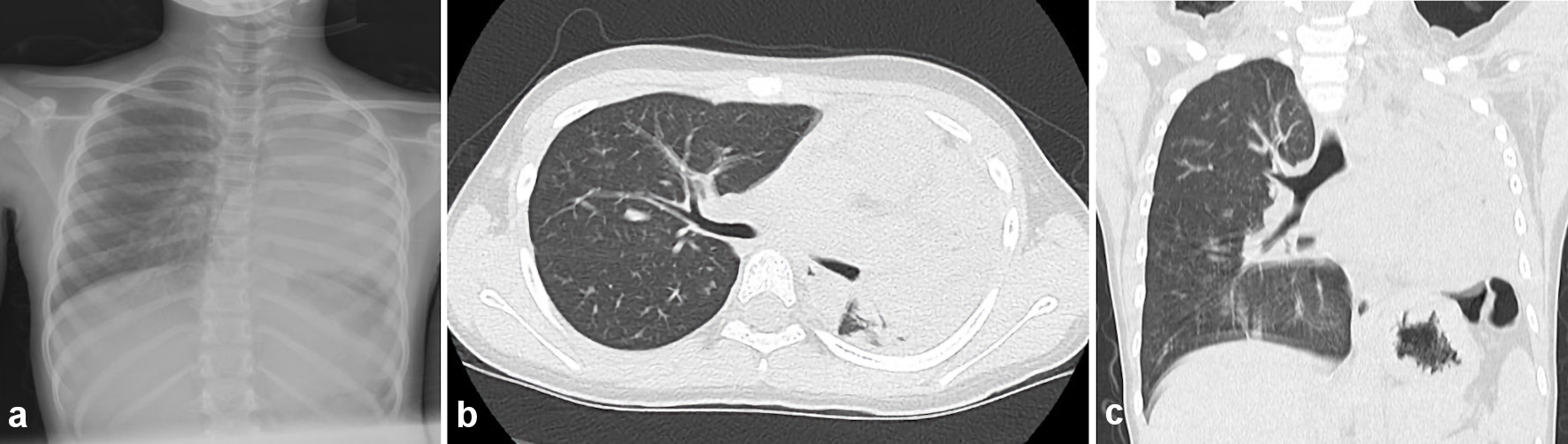 Click for large image | Figure 1. Chest radiography and computed tomography on admission day. (a) Chest radiography on admission revealed the low permeability in the left lung field. (b, c) Chest computed tomography (axial and coronal) revealed the decreasing permeability in the left lung field. |
On the next day following admission, the patient was diagnosed using a rapid antigen test for influenza (influenza A). She was diagnosed with influenza virus-induced PB and was treated with antibiotics (clarithromycin), expectant (L-carbocistein, ambroxol hydrochloride, acetylcystein inhalation), bronchodilators (procaterol hydrochloride hydrate), and antiviral drug (peramivir hydrate, 240 mg) and received intravenous infusion of steroids (methylprednisolone) (Fig. 2). The pediatric doctors explained to her parents about the bronchoscopic removal on hospital day 2. She became increasingly dyspneic, and her SpO2 was 87% (room air) despite conservative treatment via nasal oxygen (5 L/min). After supplying O2 via an oxygen mask, dyspnea was not relieved. On day 4 following admission, the patient underwent flexible bronchoscopy because of worsening dyspnea. Chest radiography and CT revealed atelectasis in the left lung field and a mucous plug in the left bronchus (Fig. 3a, b).
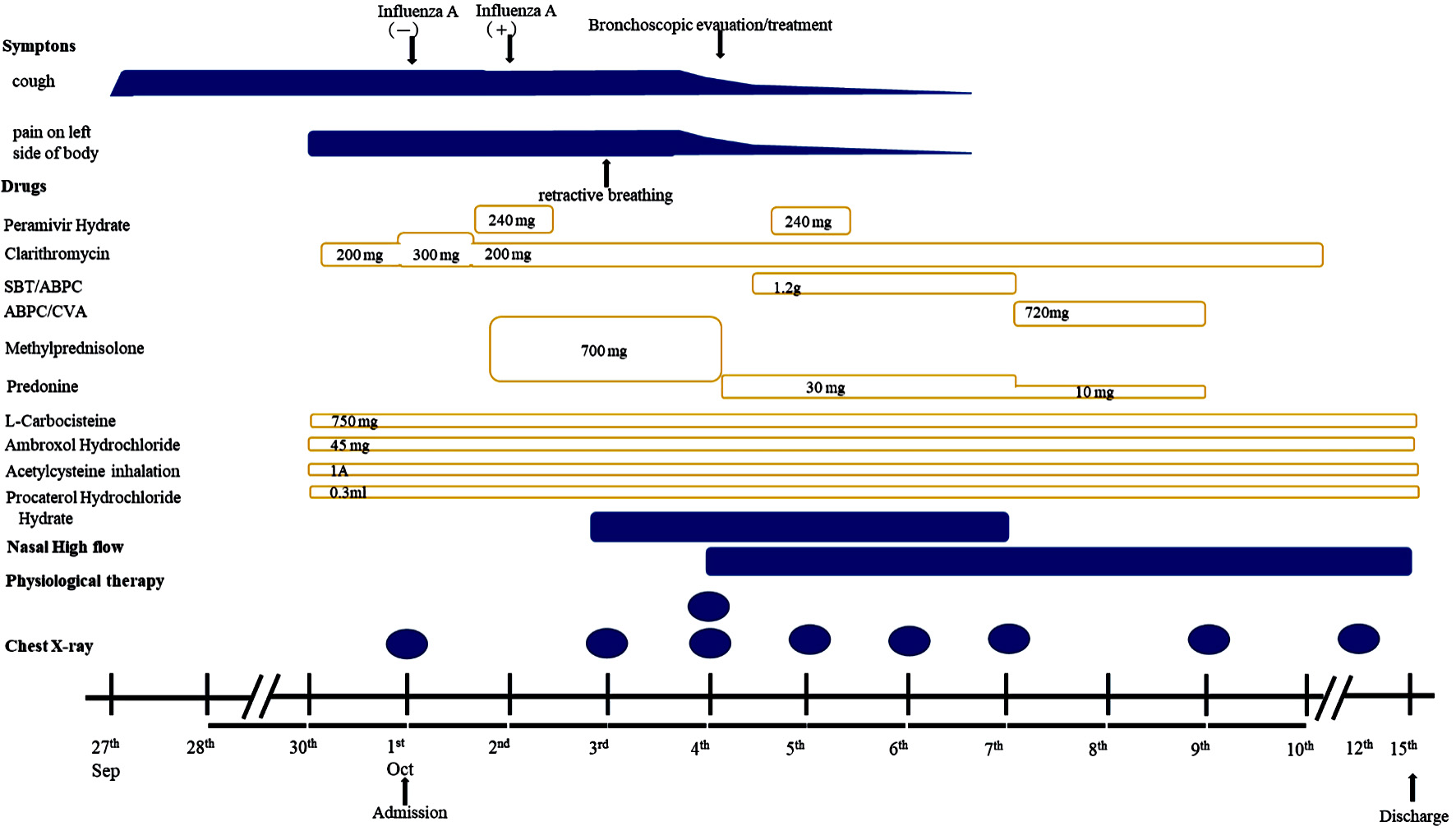 Click for large image | Figure 2. Clinical course after admission. This figure showed time course of applied medicine and chest radiography. SBT/AMPC: sulbactam/ampicillin; AMPC/CVA: amoxicillin-clavulanate. |
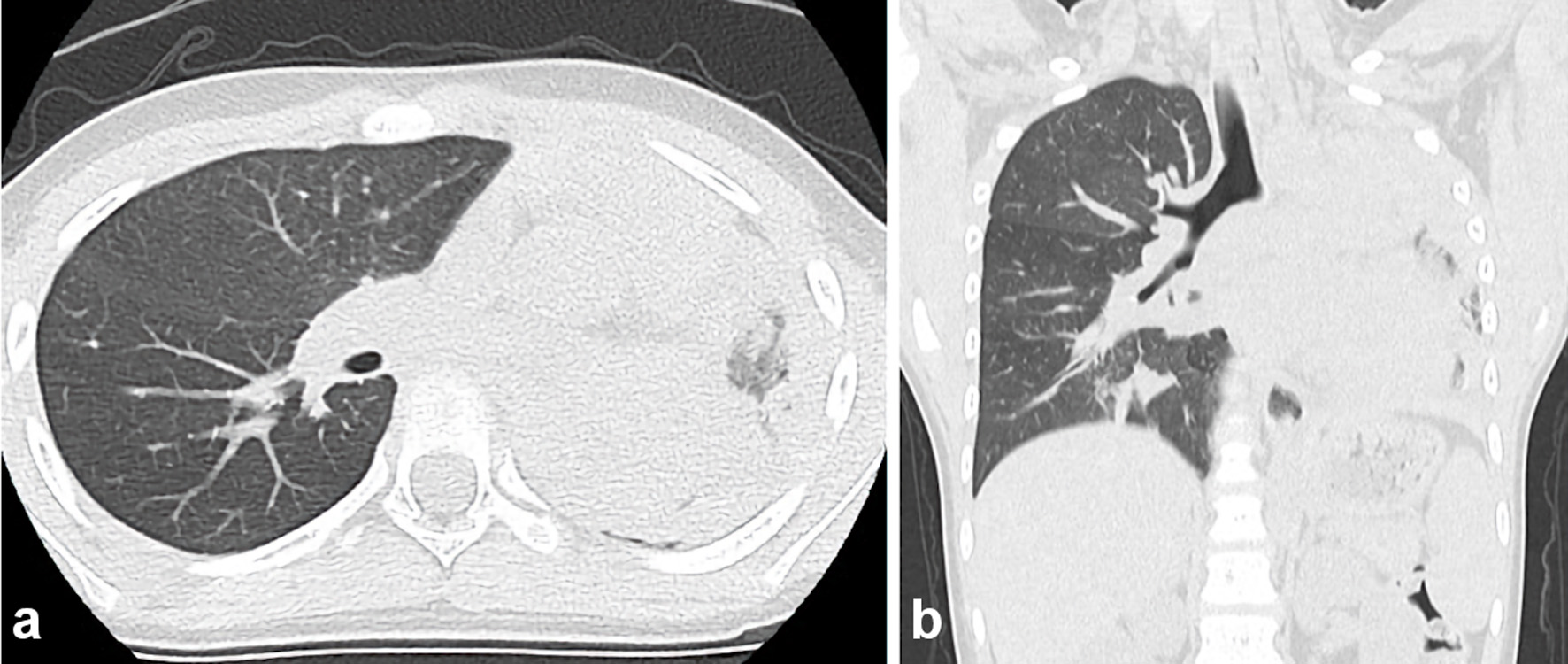 Click for large image | Figure 3. Chest computed tomography on day 4 following admission (axial and coronal) revealing the total atelectasis in the left lung field and a mucous plug in the left bronchus. |
We believed that we needed to perform flexible bronchoscopy. Flexible bronchoscopic removal of the mucosal plug was performed under general anesthesia using laryngeal mask ventilation by an anesthesiologist. A laryngeal flexible endoscope with a forceps channel was inserted, and viscous white secretions were observed in the left bronchial region. Suction was repeatedly performed (Fig. 4a-f), and the mucus plugs were removed due to cast friability and fragmentation (Fig. 5). A chest radiograph obtained after bronchoscopy and partial removal of the cast revealed marked improvement of the atelectasis finding in the left upper lung (Fig. 6a). Histopathology of the bronchial cast showed eosinophilic infiltration (Fig. 7). The patient’s left upper body pain disappeared, and shoulder breathing and mild retractive breathing improved after the bronchoscopic removal of the mucosal plug. Conservative treatment (sulbactam/ampicillin, predonine, L-carbocistein, ambroxol hydrochloride, acetylcystein inhalation, procaterol hydrochloride hydrate) was continued; she was relieved from all symptoms of the PB and atelectasis finding in her left lung on chest radiograph (Fig. 6b). She was discharged from our hospital 10 days after bronchoscopic treatment. The patient was prescribed inhaled corticosteroid (fluticasone propionate 100 µg, two times/day, for 2 years), bronchodilators (L-carbocistein 750 mg/day, and ambroxol hydrochloride 45 mg/day) and antileukotriene (pranlukast hydrate 225 mg/day, for 1 month). The patient underwent the respiratory functional test 1 month after discharge; the forced expiratory volume in 1 s (FEV1), forced vital capacity (FVC), and FEV1/FVC ratio were 1.31 L, 1.38 L, and 94.9%, respectively. The patient has been in a good condition with no respiratory infection, bronchial asthma, and recurrence of influenza virus-induced PB for 2 years after discharge.
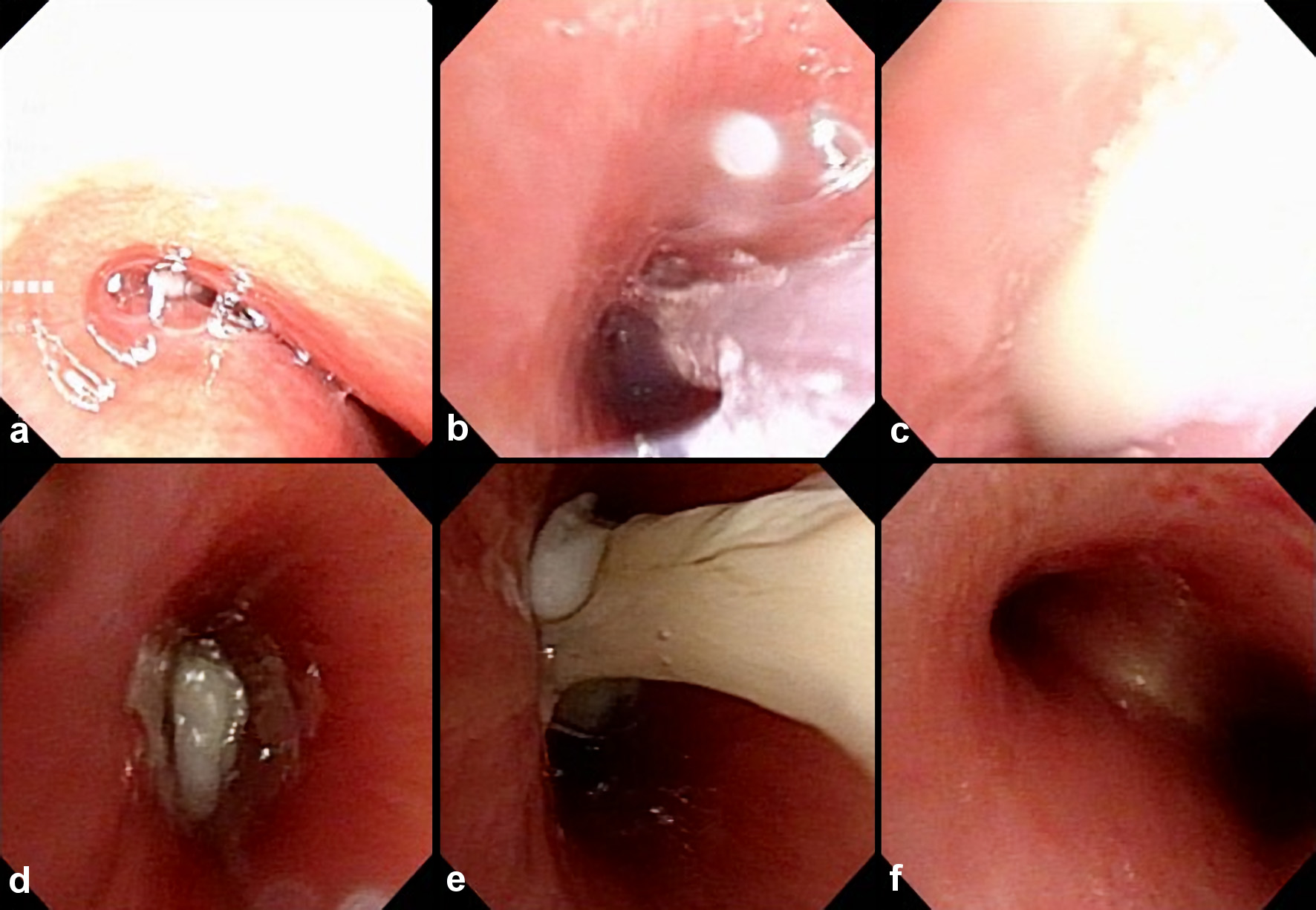 Click for large image | Figure 4. (a-f) Bronchoscopic visualization of secretions in the bronchus with patent left lobe orifice. The author (presenter, otolaryngologist) performed flexible bronchoscopy, which revealed a whitish rubbery material occluding the left lower lobe bronchus, and plastic casts were removed. The casts fragmented were removed by extraction via suctioning during fiberoptic bronchoscopy. |
 Click for large image | Figure 5. Gross findings demonstrate the fragmentation of casts. |
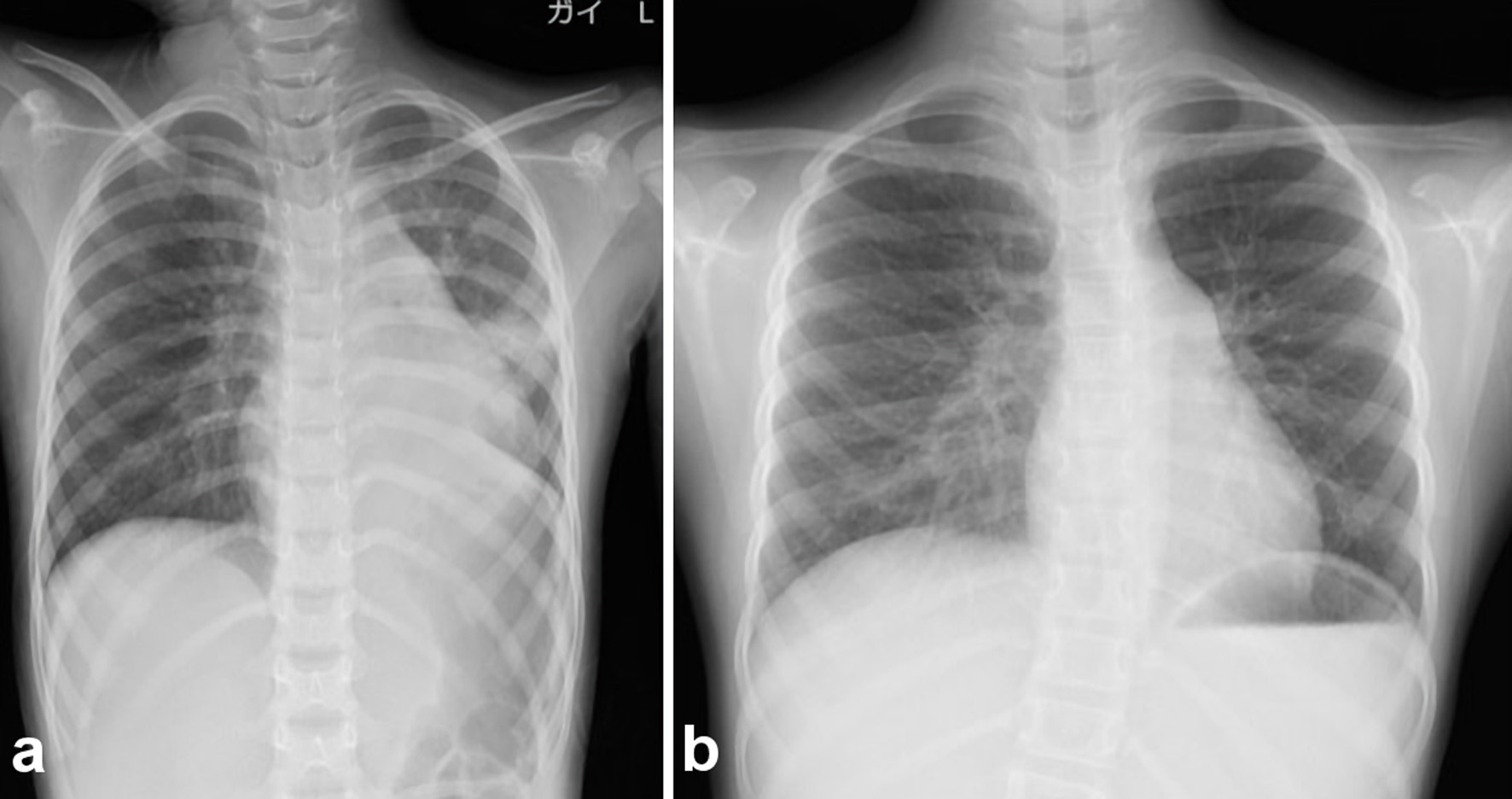 Click for large image | Figure 6. Chest radiography after bronchoscopic suctioning. (a) Finding immediately after bronchoscopic suctioning. After bronchoscopic removal of casts, the atelectasis finding in the left upper lung on chest radiography was improved. (b) Chest radiography revealed a marked improvement in the left lung. |
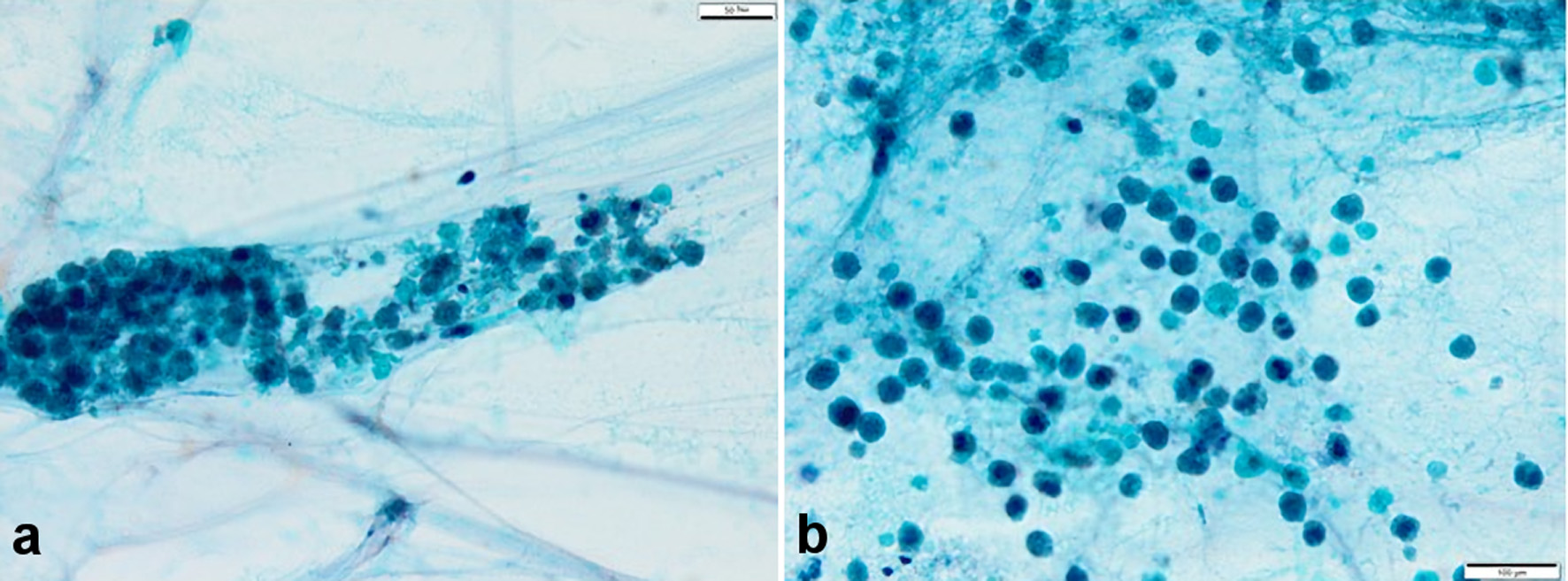 Click for large image | Figure 7. (a, b) Sputum cytopathology (hematoxylin and eosin stain, × 40). Numerous eosinophils were found; however, an evident malignant finding was not found. Eosinophils were considered the inflammatory cells. Charcot-Leyden crystals were not observed. |
This study was approved by the Faculty of Medicine Research Ethics Committee of Kyorin University (approval number, 1318).
| Discussion | ▴Top |
In this case report, we described the clinical features, chest radiographic results, and treatment for the patient with influenza virus-induced PB. A role of bronchoscopy was very important for the treatment of PB.
PB is a disease characterized by bronchial obstruction and respiratory failure due to the formation of a highly mucous plug. The earliest description of PB appeared more than 100 years previously [2]. The chief symptoms for patients with PB were dyspnea, wheezing, pleuritic chest pain, and fever [3, 4]. The typical findings of breath sounds showed poor or absent sounds in the affected lung. PB might occur more frequently in all age groups epidemiologically, and PB in children is usually associated with congenital heart disease with the Fontan procedure [5]. However, some clinicians have reported on pediatric patients with influenza virus infection. The first cases of influenza virus-induced PB were reported in an influenza A (H1N1) pdm09 pandemic that occurred in 2009. Pediatric patients with influenza virus-induced PB in the previous English language literature were reported in a total 35 cases (28 cases of influenza A [1, 6-10] and seven cases of influenza B [11-14]) (Table 2). In pediatric patients with PB-induced influenza A and influenza B, the median ages were 4 years (age, 1 year and 10 months to 11 years) and 6 years (age, 5 to 8 years), with male predominance (the ratio was 6:1 to 8:1). PB was predominantly common in women [15]; however, these differences are unknown. Interestingly, most of the patients were Asian, Japanese, Chinese, and Korean (32/35 cases, 91.4%) [1, 6-14]. Cough, wheezing, and fever, such as a common cold, were the most initial symptoms. The dyspnea was worsened, and breath sounds decreased in the affected lung.
 Click to view | Table 2. A Total 35 Cases of Pediatric Patients With Influenza Virus-Induced Plastic Bronchitis in Previous English Reports |
The most common radiographic finding was atelectasis in the affected lung, and these findings were significantly more frequent in the left lung than in the right lung [9]. However, the affected side was nonspecific (influenza A, right lung 7/28, left lung 9/28, bilateral lung 6/28, unknown side 6/28; influenza B, right lung 4/7, left lung 3/7). The underlying disease was allergy (influenza A, 11/28; influenza B, 3/7). It is likely that the underlying allergic mechanism might be a high-risk factor regardless of the type of influenza, similar with previous reports [1, 6-14].
The treatment for PB is based on the treatment of the pathologies that cause the symptoms. Common therapies include removal of mucus plugs using bronchoscopy, bronchial lavage, humidification, prescribing expectorant, and pulmonary physical therapy by a physiotherapist [11]. The treatments, including oxygen therapy, antiviral drugs (influenza A, 21/28; influenza B, 6/7), systematic steroids (influenza A, 19/28; influenza B, 5/7), and antibiotics (influenza A, 8/28; influenza B, 6/7), were dependent on the attending physician. Antiviral drugs and systemic steroids have been administered in almost all patients, but these drugs have been used based on experience. Terano et al reported that anti-influenza virus agents may not be effective for the treatment of PB [8], and Uda et al reported that antiviral drugs may improve the outcome of severe respiratory failure due to influenza A [1]. We believed that an antibiotic should be administered if the condition is pneumonia. Of the 35 patients with PB, 31 were intubated and mechanically ventilated; however, in this case report, the patient did not require intubation and mechanical ventilation. All pediatric patients underwent bronchoscopic mucosal plug removal. Bronchoscopic removal of casts was more effective when their symptoms and radiographic findings were not recovered by conservative treatment [1, 6-14]. In our case, the patient was relieved off dyspnea, shoulder breathing, and chest pain immediately after bronchoscopic intervention. However, two cases of death have been reported [10]. For a more careful point, the recurrence of the influenza virus-induced PB was reported in three patients (influenza A, two patients; influenza B, one patient) [9, 10, 14]. Interestingly, a second PB occurred due to different types of influenza virus. Recently, Mycoplasma pneumoniae infection and adenovirus-induced PB have been reported [16, 17].
We believed that the best treatment of PB was bronchoscopic removal of casts, not conservative treatment (antiviral drug, antibiotic drug, expectant, and steroid).
Conclusions
In conclusion, the possible diagnosis of PB should be considered in children with severe respiratory failure during the influenza season. In particular, allergic condition is considered a risk factor for developing PB. Bronchoscopic examination and treatment might immediately be performed after the diagnosis with PB is established.
Moreover, it is necessary to perform bronchoscopy for the differential diagnosis from an airway foreign body. We, otolaryngologists, are accustomed to using flexible endoscopes in our daily practice; therefore, we can play a role in actively performing bronchoscopic treatment, which is a treatment for PB.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Written informed consent was obtained from the patient’s mother for publication of this case report and accompanying images.
Author Contributions
M. Miyamoto contributed to conceptualization, formal analysis, writing-original draft, and writing-review and editing. Y. Yanagishita, A. Kure, S. Hashimoto and T. Takiura provided medical care. A. Hosaki provided diagnosis and medical care. K. Yan provided supervision, and K. Saito provided conceptual advice and supervision. All authors read and approved the final manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Uda K, Shoji K, Koyama-Wakai C, Furuichi M, Iwase N, Fujisaki S, Watanabe S, et al. Clinical characteristics of influenza virus-induced lower respiratory infection during the 2015 to 2016 season. J Infect Chemother. 2018;24(6):407-413.
doi pubmed - Bettmann M. Report of a fibrinous bronchitis, with a review of all cases in the literature. Am J Med Sci. 1902;123:304-329.
doi - Madsen P, Shah SA, Rubin BK. Plastic bronchitis: new insights and a classification scheme. Paediatr Respir Rev. 2005;6(4):292-300.
doi pubmed - Patel N, Patel M, Inja R, Krvavac A, Lechner AJ. Plastic bronchitis in adult and pediatric patients: a review of its presentation, diagnosis, and treatment. Mo Med. 2021;118(4):363-373.
- Caruthers RL, Kempa M, Loo A, Gulbransen E, Kelly E, Erickson SR, Hirsch JC, et al. Demographic characteristics and estimated prevalence of Fontan-associated plastic bronchitis. Pediatr Cardiol. 2013;34(2):256-261.
doi pubmed - Hasegawa M, Inamo Y, Fuchigami T, Hashimoto K, Morozumi M, Ubukata K, Watanabe H, et al. Bronchial casts and pandemic (H1N1) 2009 virus infection. Emerg Infect Dis. 2010;16(2):344-346.
doi pubmed - Deng J, Zheng Y, Li C, Ma Z, Wang H, Rubin BK. Plastic bronchitis in three children associated with 2009 influenza A(H1N1) virus infection. Chest. 2010;138(6):1486-1488.
doi pubmed - Terano C, Miura M, Fukuzawa R, Saito Y, Arai H, Sasaki M, Ariyasu D, et al. Three children with plastic bronchitis associated with 2009 H1N1 influenza virus infection. Pediatr Infect Dis J. 2011;30(1):80-82.
doi pubmed - Kim S, Cho HJ, Han DK, Choi YD, Yang ES, Cho YK, Ma JS. Recurrent plastic bronchitis in a child with 2009 influenza A (H1N1) and influenza B virus infection. J Korean Med Sci. 2012;27(9):1114-1119.
doi pubmed - Zhang J, Kang X. Plastic bronchitis associated with influenza virus infection in children: a report on 14 cases. Int J Pediatr Otorhinolaryngol. 2015;79(4):481-486.
doi pubmed - Kunder R, Kunder C, Sun HY, Berry G, Messner A, Frankovich J, Roth S, et al. Pediatric plastic bronchitis: case report and retrospective comparative analysis of epidemiology and pathology. Case Rep Pulmonol. 2013;2013:649365.
doi pubmed - Fujinaga S, Hara T. Acute kidney injury following plastic bronchitis associated with influenza B Virus in a child with nephrotic syndrome. Indian Pediatr. 2015;52(6):523-525.
doi pubmed - Nogan SJ, Cass ND, Wiet GJ, Ruda JM. Plastic bronchitis arising from solitary influenza B infection: A report of two cases in children. Int J Pediatr Otorhinolaryngol. 2015;79(7):1140-1144.
doi pubmed - Kirito Y, Matsubayashi T, Ohsugi K. Plastic bronchitis: three cases caused by influenza B virus Yamagata lineage. Pediatr Int. 2019;61(4):421-423.
doi pubmed - Eberlein MH, Drummond MB, Haponik EF. Plastic bronchitis: a management challenge. Am J Med Sci. 2008;335(2):163-169.
doi pubmed - Zhong H, Yin R, Zhao R, Jiang K, Sun C, Dong X. Analysis of clinical characteristics and risk factors of plastic bronchitis in children with mycoplasma pneumoniae pneumonia. Front Pediatr. 2021;9:735093.
doi pubmed - Yuan L, Huang JJ, Zhu QG, Li MZ, Zhuo ZQ. Plastic bronchitis associated with adenovirus serotype 7 in children. BMC Pediatr. 2020;20(1):268.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
International Journal of Clinical Pediatrics is published by Elmer Press Inc.
