| International Journal of Clinical Pediatrics, ISSN 1927-1255 print, 1927-1263 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Int J Clin Pediatr and Elmer Press Inc |
| Journal website https://www.theijcp.org |
Case Report
Volume 12, Number 1, May 2023, pages 22-27
Popliteal Artery Injury and Loss of Limb After Intraosseous Needle Placement During Resuscitation
Islam Elmitwallia, Craig Smithb, Joseph D. Tobiasa, c, d
aDepartment of Anesthesiology & Pain Medicine, Nationwide Children’s Hospital, Columbus, OH, USA
bDepartment of Orthopedic Surgery, Nationwide Children’s Hospital and The Ohio State University College of Medicine, Columbus, OH, USA
cDepartment of Anesthesiology & Pain Medicine, The Ohio State University College of Medicine, Columbus, OH, USA
dCorresponding Author: Joseph D. Tobias, Department of Anesthesiology & Pain Medicine, Nationwide Children’s Hospital, Columbus, OH 43205, USA
Manuscript submitted March 10, 2023, accepted April 26, 2023, published online May 4, 2023
Short title: IO and Vascular Damage
doi: https://doi.org/10.14740/ijcp511
| Abstract | ▴Top |
Intraosseous (IO) access is used most frequently for emergent vascular access during the care of critically ill infants and children when intravenous access cannot be rapidly achieved. Although generally safe and effective, complications may occur with extravasation of injected materials or needle damage to soft tissue, bone or vascular structures. We report the rare occurrence of damage to the popliteal artery following IO placement in the proximal tibia of an infant that resulted in distal limb ischemia and loss of limb. Although generally safe and effective, complications may occur with IO placement and its subsequent use related to extravasation of injected materials or needle damage to soft tissue, bone or vascular structures. The history of IO resuscitation is reviewed, techniques of placement discussed, and potential complications outlined.
Keywords: Intraosseous infusion; Resuscitation; Complications; Limb ischemia; Popliteal artery
| Introduction | ▴Top |
The potential clinical utility of the intraosseous (IO) route for fluid and medication administration during resuscitation was first proposed in 1922 by Drinker et al during pathologic examination of the marrow cavity of the sternum [1]. Clinical use was first reported in 12 patients with pernicious anemia who received liver concentrate administered via the marrow cavity of the sternum [2]. Use of the IO route for resuscitation was subsequently reported in 1940 by Tocantins and O’Neill who described their experience with the IO administration of blood, saline solutions, and glucose to a cohort of 17 pediatric patients [3]. During World War II, the IO route was used by the British surgeon, Dr. Hamilton Bailey, who devised a special trocar for use in the IO space of the sternum without injury to the underlying organs [4]. After the end of the second World War, use of the IO route for vascular access declined, due to the development of plastic intravenous catheters and improved intravenous cannulation techniques.
During the 1980s, the IO route for emergency vascular access re-emerged, being led by pediatric intensive care unit (ICU) physicians including Dr. James Orlowski, who described the impact of IO access on the resuscitation of severely dehydrated children during a cholera pandemic in India [5]. Subsequently, the renewed interest in IO access and continued success with its applications led to its inclusion in resuscitation guidelines from the American Heart Association and Pediatric Advanced Life Support when intravenous access was not available or could not be rapidly achieved [6]. Rapid advancement in technology has resulted in specialized devices that have increased the efficiency and accuracy of IO placement [7]. The increased experience with the technique and improvements in technology have led to the suggestion that IO access may have a role not only in emergent, but also semi-elective scenarios when intravenous access fails or is not technically feasible [8].
Although generally safe and effective, complications may occur with IO placement and its subsequent use related to extravasation of injected materials or needle damage to soft tissue, bone or vascular structures. We report damage to the popliteal artery following IO placement in the proximal tibia of an infant that resulted in distal limb ischemia. Clinical uses of IO resuscitation are reviewed, techniques of placement discussed, and potential complications outlined.
| Case Report | ▴Top |
Review of this case and presentation in this format followed the guidelines of the Institutional Review Board of Nationwide Children’s Hospital (Columbus, Ohio). The patient was a 2-month-old infant who presented to an outside hospital for treatment of vomiting, dehydration, and weight loss. Past history was positive preterm birth at 35 weeks gestation, intrauterine growth retardation, poor feeding, and jaundice requiring a 2-week admission to the neonatal ICU. Following discharge from the neonatal ICU stay, she had been doing well and had gained weight up to approximately 4 kg. A clinical diagnosis of presumed viral gastroenteritis was made and she was admitted to pediatric inpatient ward for additional workup and management of dehydration. Following hospital admission, multiple attempts to gain peripheral venous vascular access were unsuccessful. Therefore, the decision was made to provide enteral nutrition and rehydration through a nasogastric tube. Workup for the gastroenteritis revealed an adenovirus infection. During the continued enteral feeding, the patient continued to have diarrhea with persistent metabolic acidosis. Further workup for an inborn error or metabolism or renal tubular acidosis was negative. She also developed a urinary tract infection (Klebsiella pneumoniae) during her inpatient hospitalization which was treated with a 7-day course of intravenous ceftriaxone. She had a prolonged 2-week inpatient hospital course without significant improvement. Subsequently, she had an acute episode of altered mental status with lethargy and agonal respirations that progressed to cardiac arrest with pulseless electrical activity. She was transferred to the pediatric ICU and her trachea was intubated. During the resuscitation, an IO needle was placed in the left tibia. Following two doses of epinephrine, 30 mL/kg of isotonic fluids, and 11 min of cardiopulmonary resuscitation, there was return of spontaneous circulation. The IO access lasted only temporally with swelling noted in the area surrounding the insertion site and no free flow of fluid. The IO needle was removed. Other attempts to gain vascular access were unsuccessful, including IO access into the right proximal tibia and central vascular access into both femoral veins and the right internal jugular vein. To secure venous access, the patient was transferred to the operating room, a venous cut-down performed, and a Broviac® catheter placed into the left external jugular vein. Further evaluation to determine the etiology of the cardiac arrest revealed hypoglycemia with a blood glucose of 16 mg/dL at the time of cardiac arrest. Following the cardiac arrest, she developed acute kidney injury (peak serum creatinine 1.49 mg/dL) and computed tomography (CT) of the head revealed a right transverse and sigmoid dural venous sinus thrombosis. Following the resuscitation, there was decreased perfusion and cyanosis of the left lower extremity with significant color change from the mid-tibia distally to the foot (Fig. 1). Arterial thrombosis was suspected and a heparin infusion was started. Arterial duplex ultrasound displayed lack of flow in the popliteal artery distal to the popliteal fossa. The patient was then transferred to our hospital for further assessment and management. Initial physical examination revealed stable vital signs with a blood pressure of 74/40 mm Hg and heart rate of 140 - 150 beats/min. The trachea was intubated and ventilation controlled. The infant was alert, slightly hypotonic, but prior to sedation, was moving all extremities spontaneously although somewhat decreased on the left lower extremity. There was symmetric facial movement and the pupils were equal, round, and reactive. Vascular surgery consultation was of the opinion that the limb was non-salvageable and the patient was not a surgical candidate due to her size, age, associated renal and cardiac involvement, and given that the injury had occurred more than 6 h ago. During her subsequent hospitalization, there was a decrease in the platelet count to 47,000/mm3 which prompted an evaluation for heparin-induced thrombocytopenia (HIT) and a transition to argatroban for systemic anticoagulation due to the dural venous thrombosis. The HIT panel was subsequently negative, the thrombocytopenia resolved (was attributed to ongoing disseminated intravenous coagulation following the cardiac arrest), and heparin was restarted. The patient was subsequently weaned from mechanical ventilation and her trachea extubated on day 4 of her admission to our hospital. The affected limb showed progressive demarcation of the injured extremity (Fig. 2). On day 11, the patient underwent a through the knee amputation of the left extremity. The remainder of her hospital course was uneventful. Renal function returned to normal. There were no apparent neurologic sequelae from the cardiac arrest and the dural vein thrombosis. She has subsequently been fitted with a prothesis and is doing well at 17 months of age with outpatient follow-up with neurology, orthopedic surgery, and gastroenterology services.
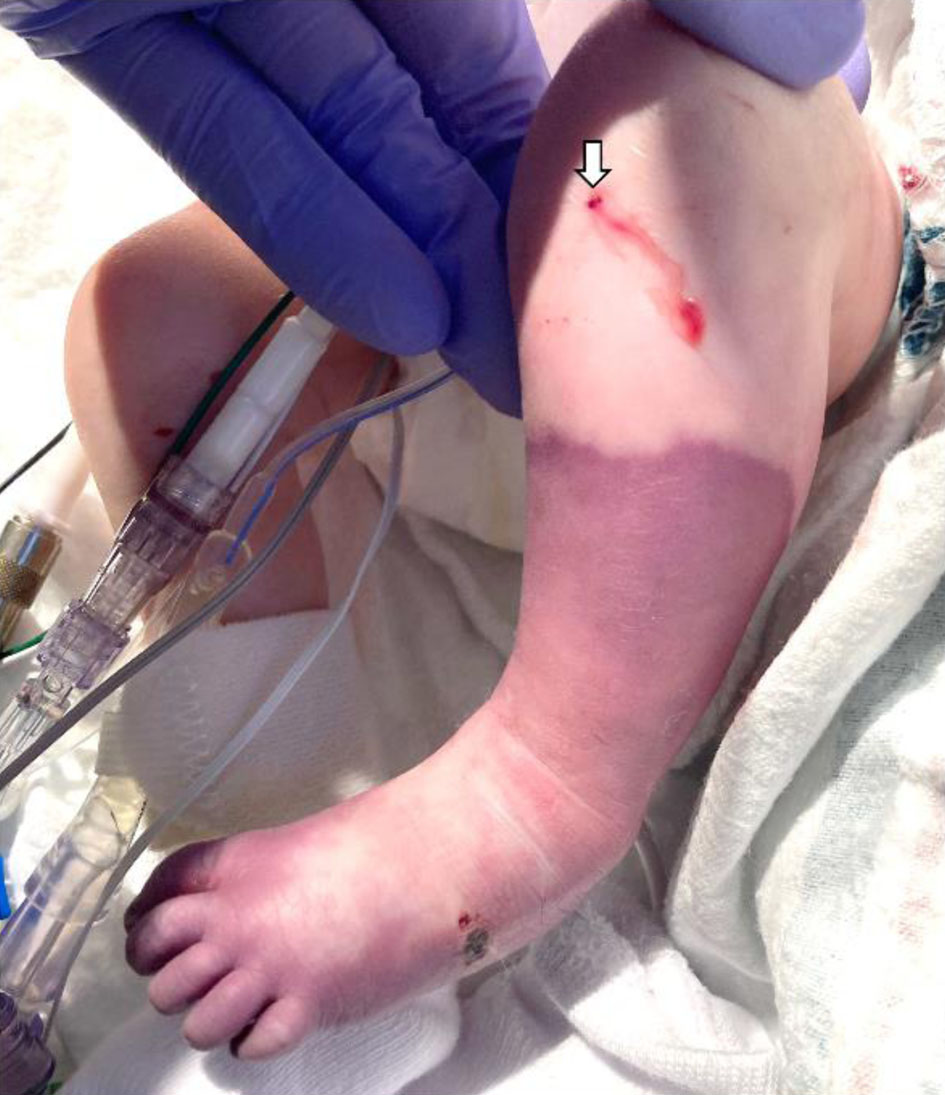 Click for large image | Figure 1. Following the resuscitation, there was progressive decreased perfusion and cyanosis of the left lower extremity with significant color change from the mid-tibia distally to the foot. The intraosseous needle insertion site is noted (white arrow). |
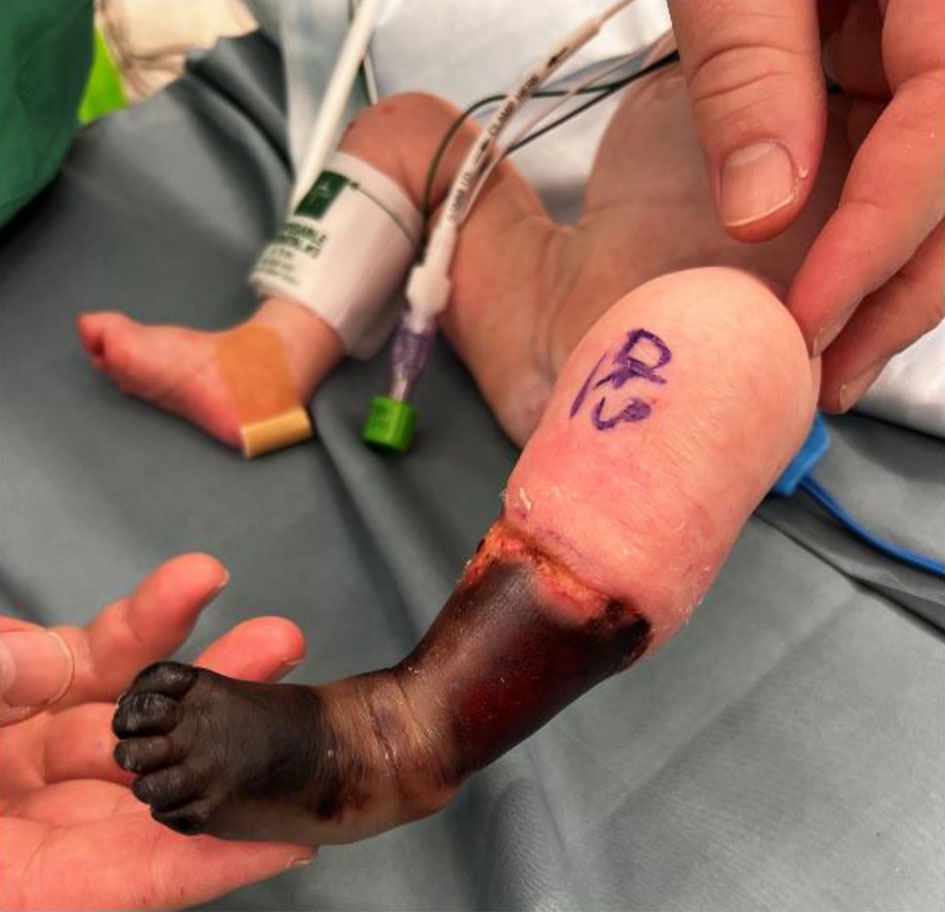 Click for large image | Figure 2. Photograph showing progressive demarcation of the injured extremity. |
| Discussion | ▴Top |
The IO route continues to be the preferred route of access and medication administration during resuscitation when intravenous access is not present or cannot be rapidly obtained within three attempts or 90 s. Although IO access is generally safe and effective with a low incidence of complications, complications may occur during initial needle placement or during the administration of medications. Serious adverse events including lower limb ischemia have been anecdotally reported especially in neonates and small infants.
Although the first use of IO access involved insertion of the needle into the intramedullary cavity of the sternum of adults, the practice has changed primarily to involve the use of the intramedullary cavity of long bones, primarily the proximal tibia [9, 10]. Since its reintroduction into clinical practice in the 1980s, IO access has been shown to be superior to or equivalent to intravenous access for resuscitation in both hospital and pre-hospital settings [11-14].
Despite the relative safety of the technique, significant complications have been reported, related either to the initial placement of the needle or its subsequent use. Complications related to initial needle placement include osteomyelitis, bone fracture, superficial cellulitis, osteomyelitis, misplacement of the needle with tissue or vascular damage, and injury to the growth plate. The latter are most common when needle placement is too high on the proximal tibia as opposed to the recommended 1 - 2 cm inferior and medial to the tibial tuberosity on the medial face of the tibia. Complications may occur related to the extravasation of fluid and medications including tissue damage and necrosis, the development of compartment syndrome, or even loss of limb. We postulate that the complication noted in our patient resulted from the needle penetrating the posterior aspect of the tibia and directly damaging the popliteal artery during its use for the initial resuscitation. Alternatively, extravasation of fluid or medications may have resulted in damage to and thrombosis of the popliteal artery.
Direct vascular injury may occur anywhere along the course of the artery. In our case, we postulate that the IO needle penetrated the posterior aspect of the tibia and directly damaged the popliteal artery as it exits the popliteal fossa (Fig. 3). The femoral artery originates from the external iliac artery and travels anterior in the proximal thigh. It then enters the adductor canal in the medial midthigh and then crosses posterior behind the medial femoral condyle. There, it transitions into the popliteal artery as it enters the popliteal fossa and heads posterior to the proximal tibia.
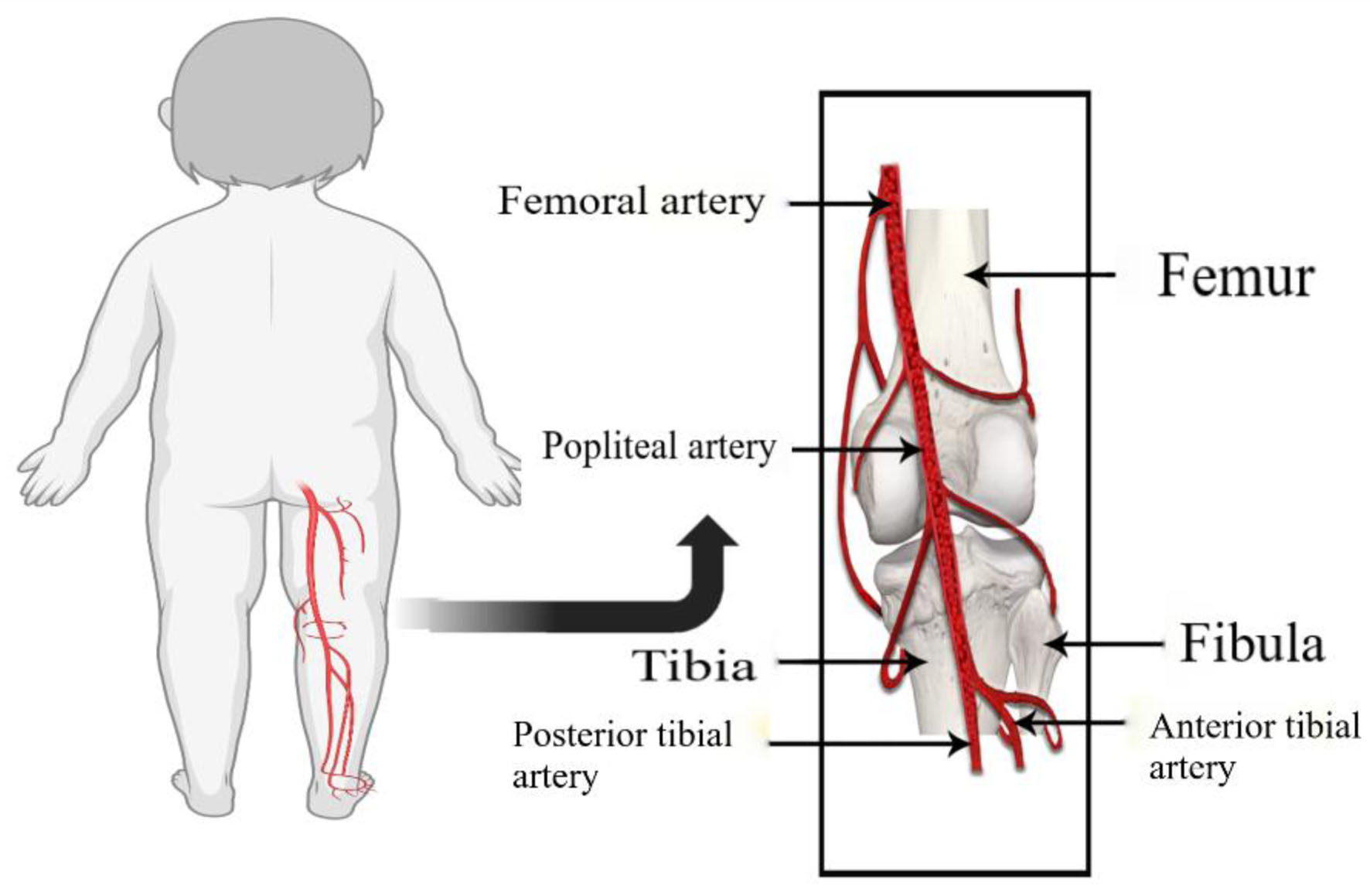 Click for large image | Figure 3. Diagram showing the course of the popliteal artery. The femoral artery, a continuation of the external iliac artery travels through the adductor canal on the medial side of the femur above the medial epicondyle. The adductor canal, an opening in the adductor magnus muscle hiatus, is located at the junction of the middle and lower thirds of the thigh. The artery is anterior to the femur in the femoral canal, but then travels anteromedially, and crosses behind the medial epicondyle of the femur into the popliteal fossa. In the popliteal fossa, it becomes the popliteal artery and travels posterior to the tibia. |
Initial misplacement of the needle or its dislodgment during subsequent use can lead to the extravasation of fluid and medications. Regardless of the fluid administered, the extravasation of large volumes into limited tissue spaces can result in the development of compartment syndrome. Various reviews have focused on potential complications and their incidence during IO placement and its subsequent use [15-17]. In a retrospective review of the electronic health record data base involving 1,012 patients from 37 institutions, complications were noted in 29 pediatric patients for an incidence of 2.9% [15]. Lower limb ischemia was the most commonly reported adverse event, occurring in 16 patients (1.6%), most of whom (n = 13) were less than 1 year of age. A subsequent study used an on-line questionnaire to accumulate data from physicians’ recollections of clinical IO use [16]. A total of 1,802 clinical cases of IO use was reported by 386 responders. Reported problems with establishing IO access included inability to aspirate bone marrow (12.3%), difficulties with penetration of the periosteum with the IO needle (10.3%), patient discomfort/pain (7.1%), and a bent of broken needle (4.0%). When using an established IO access, the reported complications were difficulties with injection of fluid and medications (7.4%), slow infusion (8.8%), movement and dislodgement after insertion (8.5%), and extravasation of fluid (3.7%). More serious complications included compartment syndrome (0.6%) and osteomyelitis (0.4%). Although the majority of these complications can be identified early and treated by discontinuation of the IO infusion, anecdotal reports have documented potentially devasting injuries including loss of limb [18-22].
A key to the prevention of injury is use of the correct technique and equipment. This is especially true in neonates and infants as the majority of reports of severe limb ischemia and compartment syndrome following IO access have been in this patient population. In neonates, given the limited width of the intramedullary cavity in neonates (approximately 7 mm in a neonate and 10 mm in an infant), there is an increased risk that the needle may penetrate the entire intramedullary cavity and exit the posterior cortical side especially with repeated attempts of needle insertions in the same location. The needle should be inserted 1 - 2 cm below the tibial tuberosity and 1 cm medially toward the tibial plateau. This point is preferred as the cortex of the tibia is the thinnest and the adjacent epiphyseal cartilage plate can be avoided. Insertion of the IO needle should be perpendicular to the skin. The needle is advanced until loss of resistance is felt, which indicates needle passage through the cortex and positioning within the intramedullary space. At this point, the needle should hold itself in position without external stabilization. Further confirmation can occasionally be achieved by aspiration of blood or marrow elements. Most importantly, there should be free flow of fluid by gravity. Saline should be able to be injected with a syringe without resistance or signs of extravasation. If either occur, the needle should be removed and reinserted in a different extremity. Early detection of extravasation may help in preventing the rare and severe complications of limb ischemia and compartment syndrome. Recently, the use of ultrasound has been suggested as an additional means of confirming placement by real-time imaging of the IO space during infusion of a small volume of fluid. Absence of Doppler colored signals within the IO space indicates extravasation of fluid to the surrounding structures [23]. Ongoing assurance of the correct location of the IO needle is mandatory not only for its initial use but also during the ongoing resuscitation as the needle may become dislodged during prolonged resuscitations with cardiopulmonary resuscitation (CPR) and patient movement.
Use of the correct equipment can also decrease the potential for complications by ensuring correct needle location. There are a wide variety of commercially available needles and devices [24]. Although initial clinical experience used a wide variety of readily accessible needles including bone marrow biopsy needles, spinal needles, and standard intravenous venous, there are now several specialty devices for IO access. These include semi-automatic spring-loaded devices like the Bone Injection Gun (BIG™), New Intraosseous Device (NIO™), and the First Access for Shock and Trauma 1 (FAST-1™). There are also battery-driven drill devices such as the Easy Intraosseous Access Device (EZ-IO™) [7, 25, 26]. The latter device has needles of various lengths (15, 25, and 45 mm), which are chosen based on the patient’s age, weight, and site of IO access.
The IO route is mostly commonly used for the administration of resuscitation medications (epinephrine, atropine) or isotonic fluids (normal saline). However, it can be used for administration of several other medications including amiodarone, dopamine, fentanyl, ketamine, neuromuscular blocking agents (rocuronium, succinylcholine), antibiotics, and electrolytes. Administration of hypertonic fluids (greater than 0.9% saline, glucose concentrations greater than 10%), strongly alkaline (sodium bicarbonate), or potentially caustic solutions (calcium) is not recommended [27]. Additionally, care should be exercised when medications, which may result in vasoconstriction and tissue necrosis (norepinephrine), are infused into the IO space. Appropriate equipment and assurance of correct needle location in the IO space are mandatory to limit the incidence of complications. The IO access should be immediately removed once suitable intravenous access is obtained. With these caveats in mind, IO access can be life-saving during resuscitative efforts in both pediatric and adult patients.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Informed consent was obtained from a parent for anesthetic care and use of de-identified information for publication.
Author Contributions
IE performed the initial case review and manuscript preparation, literature review, and editing of subsequent revisions. JDT provided clinical care, participated in the literature review, and edited the manuscript. CS provided clinical care and participated in preparation of the manuscript including editing of the final version.
Data Availability
The data supporting the findings of this case report are available from the authors.
| References | ▴Top |
- Drinker CK, Drinker KR, Lund CC. The circulation in the mammalian bone marrow. Am J Physiol. 1922;62:1-92.
- Josefson A. A new method of treatment - intraosseous injection. Acta Med Scand. 1934;81:550-554.
- Tocantins LM, O’Neill JF. Infusion of blood and other fluids into the circulation via the bone marrow. Proc Soc Exp Biol Med. 1940;45:782-783
- Bailey H. Bone-marrow injections. Br Med J. 1944;1(4335):181-182.
doi pubmed pmc - Orlowski JP. My kingdom for an intravenous line. Am J Dis Child. 1984;138(9):803.
doi pubmed - Tobias JD, Ross AK. Intraosseous infusions: a review for the anesthesiologist with a focus on pediatric use. Anesth Analg. 2010;110(2):391-401.
doi pubmed - Gazin N, Auger H, Jabre P, Jaulin C, Lecarpentier E, Bertrand C, Margenet A, et al. Efficacy and safety of the EZ-IO intraosseous device: Out-of-hospital implementation of a management algorithm for difficult vascular access. Resuscitation. 2011;82(1):126-129.
doi pubmed - Neuhaus D, Weiss M, Engelhardt T, Henze G, Giest J, Strauss J, Eich C. Semi-elective intraosseous infusion after failed intravenous access in pediatric anesthesia. Paediatr Anaesth. 2010;20(2):168-171.
doi pubmed - Chreiman KM, Dumas RP, Seamon MJ, Kim PK, Reilly PM, Kaplan LJ, Christie JD, et al. The intraosseous have it: A prospective observational study of vascular access success rates in patients in extremis using video review. J Trauma Acute Care Surg. 2018;84(4):558-563.
doi pubmed pmc - Granfeldt A, Avis SR, Lind PC, Holmberg MJ, Kleinman M, Maconochie I, Hsu CH, et al. Intravenous vs. intraosseous administration of drugs during cardiac arrest: A systematic review. Resuscitation. 2020;149:150-157.
doi pubmed - Besserer F, Kawano T, Dirk J, Meckler G, Tijssen JA, DeCaen A, Scheuermeyer F, et al. The association of intraosseous vascular access and survival among pediatric patients with out-of-hospital cardiac arrest. Resuscitation. 2021;167:49-57.
doi pubmed - Recher M, Baert V, Escutnaire J, Le Bastard Q, Javaudin F, Hubert H, Leteurtre S. Intraosseous or peripheral IV access in pediatric cardiac arrest? Results from the French national cardiac arrest registry. Pediatr Crit Care Med. 2021;22(3):286-296.
doi pubmed - Reades R, Studnek JR, Vandeventer S, Garrett J. Intraosseous versus intravenous vascular access during out-of-hospital cardiac arrest: a randomized controlled trial. Ann Emerg Med. 2011;58(6):509-516.
doi pubmed - Orlowski JP, Porembka DT, Gallagher JM, Lockrem JD, VanLente F. Comparison study of intraosseous, central intravenous, and peripheral intravenous infusions of emergency drugs. Am J Dis Child. 1990;144(1):112-117.
doi pubmed - Hoskins M, Sefick S, Zurca AD, Walter V, Thomas NJ, Krawiec C. Current utilization of interosseous access in pediatrics: a population-based analysis using an EHR database, TriNetX. Int J Emerg Med. 2022;15(1):65.
doi pubmed pmc - Hallas P, Brabrand M, Folkestad L. Complication with intraosseous access: scandinavian users' experience. West J Emerg Med. 2013;14(5):440-443.
doi pubmed pmc - Konopka E, Webb K, Reserva J, Moy L, Ton-That H, Speiser J, Tung R. Cutaneous complications associated with intraosseous access placement. Cutis. 2021;107(6):E31-E33.
doi pubmed - Tareq AA. Gangrene of the leg following intraosseous infusion. Ann Saudi Med. 2008;28(6):456-457.
doi pubmed pmc - Suominen PK, Nurmi E, Lauerma K. Intraosseous access in neonates and infants: risk of severe complications - a case report. Acta Anaesthesiol Scand. 2015;59(10):1389-1393.
doi pubmed - Gayle M, Kissoon N. A case of compartment syndrome following intraosseous infusions. Pediatr Emerg Care. 1994;10(6):378.
pubmed - Molacek J, Houdek K, Opatrny V, Fremuth J, Sasek L, Treskova I, Treska V. Serious complications of intraosseous access during infant resuscitation. European J Pediatr Surg Rep. 2018;6(1):e59-e62.
doi pubmed pmc - Launay F, Paut O, Katchburian M, Bourelle S, Jouve JL, Bollini G. Leg amputation after intraosseous infusion in a 7-month-old infant: a case report. J Trauma. 2003;55(4):788-790.
doi pubmed - Abramson TM, Alreshaid L, Kang T, Mailhot T, Omer T. FascIOtomy: Ultrasound Evaluation of an Intraosseous Needle Causing Compartment Syndrome. Clin Pract Cases Emerg Med. 2018;2(4):323-325.
doi pubmed pmc - Drozd A, Wolska M, Szarpak L. Intraosseous vascular access in emergency and trauma settings: a comparison of the most universally used intraosseous devices. Expert Rev Med Devices. 2021;18(9):855-864.
doi pubmed - Dolister M, Miller S, Borron S, Truemper E, Shah M, Lanford MR, Philbeck TE. Intraosseous vascular access is safe, effective and costs less than central venous catheters for patients in the hospital setting. J Vasc Access. 2013;14(3):216-224.
doi pubmed - Davidoff J, Fowler R, Gordon D, Klein G, Kovar J, Lozano M, Potkya J, et al. Clinical evaluation of a novel intraosseous device for adults: prospective, 250-patient, multi-center trial. JEMS. 2005;30(10):S20-S23.
pubmed - Alam HB, Punzalan CM, Koustova E, Bowyer MW, Rhee P. Hypertonic saline: intraosseous infusion causes myonecrosis in a dehydrated swine model of uncontrolled hemorrhagic shock. J Trauma. 2002;52(1):18-25.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
International Journal of Clinical Pediatrics is published by Elmer Press Inc.
